The Trump Administration Just Moved Marijuana from Schedule I to Schedule III: Here’s What That Actually Means.
The Trump administration has reclassified medical marijuana from Schedule I to Schedule III. This is a meaningful shift in federal policy, but it is not legalization, and it is not a green light for recreational use. It is the federal government acknowledging that cannabis has medical value and should not sit in the same category as heroin and LSD.
The practical question is what this actually changes. Read on to find out.
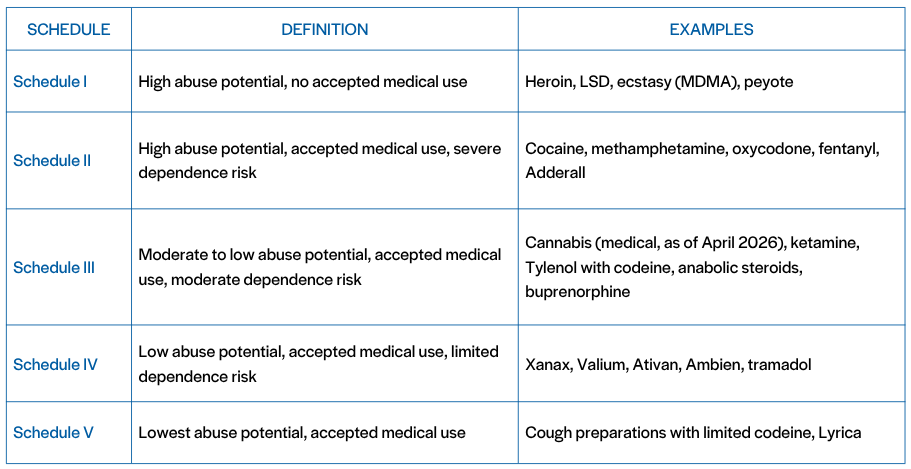
What is Schedule I? And What is Schedule III?
Schedule I is for substances with high abuse potential and no accepted medical use. Schedule III recognizes accepted medical use and lower abuse potential. Cannabis now sits alongside ketamine and codeine-containing products (Schedule I), not heroin (Schedule III).
The change applies immediately to FDA-approved marijuana products and to medical marijuana regulated under qualifying state-issued licenses, per the Department of Justice. Broader rescheduling, including non-medical marijuana, goes to a DEA administrative hearing beginning June 29, 2026. The immediate scope is narrower than most coverage implies.
What This Changes Medically
Schedule I status created real barriers to research and limited what could be studied about safety and dosing. Schedule III eases those barriers and allows for more rigorous clinical study, as PBS reported.
This opens up dosing data, drug interaction studies, route-of-administration research, and long-term safety evidence, all of which have lagged behind actual patient use.
Reclassification is not an endorsement of cannabis as harmless. It is still a drug with physiologic effects, side effects, and contraindications, and it should be used in the right clinical context.
What This Changes for Patients
Many patients already use cannabis for pain, sleep, anxiety, and nausea. Schedule III supports more open conversations between patients and providers, which produces safer and more individualized recommendations.
This is not a blanket endorsement of every cannabis product on the market. Quality, potency, labeling, and route of administration vary widely. The clinical conversation stays anchored in safety, evidence, and appropriate use.
What This Changes at the State Level
State law still drives what patients actually experience. Federal reclassification does not override state cannabis laws or create a uniform national policy.
For states with medical cannabis programs, this strengthens program legitimacy and reduces friction with federal policy. Expect downstream effects on taxation, licensing, banking access, and how state programs interface with federal rules. Adult-use cannabis is a separate conversation, and broader change depends on the June 2026 hearing and the rulemaking that follows.
What This Changes for Recreational Use
Not much, practically speaking. This is not nationwide legalization. It does not erase the distinction between medical and recreational markets, and it does not remove federal restrictions on adult-use cannabis.
What it does do is move cannabis closer to mainstream federal acceptance and reshape the policy conversation. Rescheduling and legalization are not the same thing.
The Bigger Picture of Marijuana Re-Classification
This is a step, not an endpoint. Federal cannabis policy has been out of sync with clinical reality and patient use for years, and this rescheduling closes part of that gap. It creates room for better research, better clinical guidance, and clearer conversations between patients and providers.
Marijuana has not been legalized. It has been reclassified in a way that acknowledges medical relevance, and the implications for healthcare, policy, and patient care will continue to unfold as the June hearing and broader rulemaking move forward.