What Research on IBD and Depression Tells Us about the Gut-Brain Connection
For a long time, the thinking went like this: if someone has a chronic illness, of course they might also be depressed. Living with pain or fatigue or unpredictable symptoms is hard. Depression was treated as a reasonable emotional response to a difficult physical situation.
The research of chronic illness and depression has evolved. A review published in April 2026 in Frontiers in Pharmacology pulls together years of evidence on depression in people with inflammatory bowel disease (IBD), and the picture it paints is not a one-way street. The research shows how the gut and the brain are talking to each other constantly, and when that conversation breaks down, both ends suffer. The symptoms we call depression and the symptoms we call gut disease can actually be two expressions of the same underlying problem.
This is relevant for more than IBD. The same loop shows up in chronic pain, anxiety, and a range of conditions that cross the line between physical and psychiatric. Understanding how it works changes how we think about treatment.
This is why, at our practice, we treat the body as one conversation. What shows up as mood and what shows up as pain (or gut discomfort) are often the same signal, speaking in two languages.
The Gut-Brain Loop, In Plain Terms
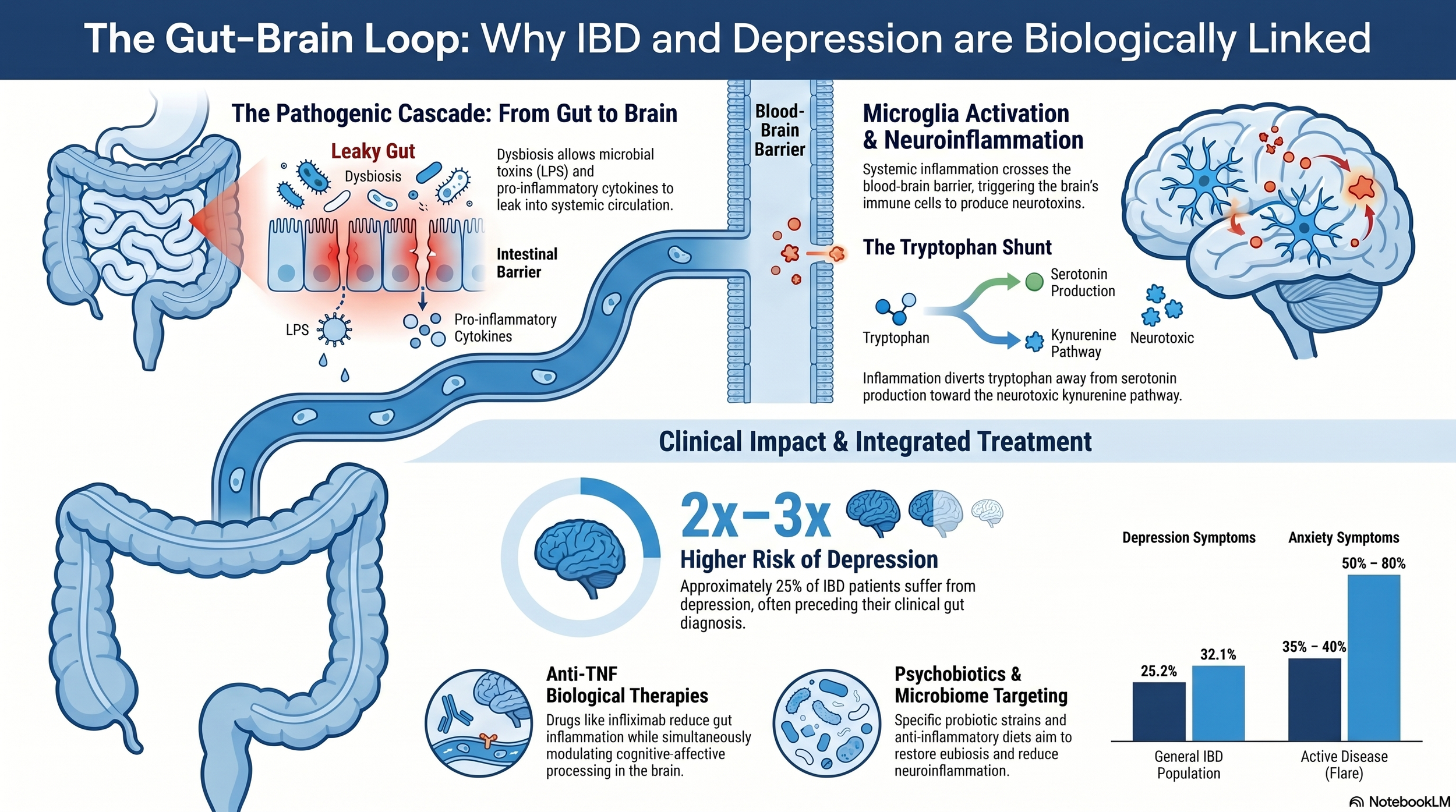
Start with the gut. Inside it, trillions of microorganisms live in a balance that, when healthy, helps digest food, regulate immunity, and produce signaling molecules that affect mood and pain. When that balance shifts (a state called dysbiosis), a few things happen in sequence:
Beneficial bacteria that produce butyrate, a short-chain fatty acid that fuels the cells lining the colon, get depleted.
This depletion causes the gut lining to weaken.
Bacterial components, particularly one called lipopolysaccharide (LPS), start leaking into the bloodstream.
The immune system, recognizing these components as foreign, activates and releases inflammatory messengers called cytokines (TNF-alpha, IL-6, IL-1beta, among others).
These cytokines do not stay in the gut. They travel through the bloodstream, increase the permeability of the blood-brain barrier, and activate microglia, the immune cells that live in the brain.
Once activated, microglia start producing their own inflammatory signals inside the brain. That state is called neuroinflammation.
Neuroinflammation changes how the brain works. It affects how tryptophan, the precursor to serotonin, gets processed. Under normal conditions, tryptophan feeds into serotonin production. Under inflammatory conditions, it gets diverted into a different pathway that produces neurotoxic metabolites, including one called quinolinic acid, which contributes to excitotoxicity and damage in the hippocampus.
Serotonin levels drop. Neuroplasticity suffers. The symptoms we recognize as depression start to emerge.
That is the gut-to-brain direction. But the loop runs the other way too.
The Brain Talks Back to the Gut
Stress and anxiety activate the HPA axis, the body's central stress response system. The result is elevated cortisol. In short bursts, cortisol is anti-inflammatory and useful. In chronic elevation, the body develops cortisol resistance, which flips cortisol's effect. Tissues stop responding to its anti-inflammatory signals, and a pro-inflammatory state takes hold instead.
Chronic cortisol also damages the intestinal barrier directly. It reduces mucus secretion and weakens the tight junctions between cells in the gut lining. That makes the gut more permeable. More LPS leaks out. More cytokines get released. More inflammation reaches the brain.
So the loop closes. Gut inflammation drives brain inflammation. Brain stress drives gut inflammation. Neither starts things independently; they keep each other going.
The Gut-Brain Axis in IBD
IBD (Crohn's disease and ulcerative colitis) is a condition in which this loop is especially visible. The gut inflammation is severe and chronic by definition. What the research now shows is that the psychiatric symptoms that accompany IBD are not incidental.
Across pooled studies, about 23-25% of people with IBD have depressive symptoms. About 32% have anxiety symptoms. During active flares, those rates climb to 35-40% for depression and up to 80% for anxiety. That is two to three times the rate seen in the general population.
The direction of the relationship is also telling. An IBD diagnosis raises the risk of later developing depression by about 55%. But a prior diagnosis of depression raises the risk of later developing IBD by 20 to 100 percent.
Depression, in other words, often precedes IBD. This rules out the old model where depression is just a reaction to having a hard disease. The two are sharing biology from the start.
Comorbid depression in IBD also makes the IBD itself worse. People with both face more flares, more hospitalizations, more surgeries, and weaker response to the standard biologic treatments. Treating one without the other leaves a lot of the picture untouched.
How Interventions Can Support Both the Gut and the Brain
Because of this loop, interventions at any point in the loop can shift the whole system. The review pulls together evidence on several treatment approaches, and a few patterns stand out.
Biologic medications that target inflammation (particularly anti-TNF drugs like infliximab and adalimumab) have measurable antidepressant effects in people with IBD. Brain imaging has shown changes in the left amygdala, right prefrontal cortex, and posterior cingulate cortex, regions involved in mood regulation and self-perception (Gray et al., 2018). Head-to-head comparisons found anti-TNF drugs produced significantly larger reductions in depressive symptoms than other immunomodulators in this population (Horst et al., 2015). These drugs were designed for the gut, but they are acting on the brain too.
In the other direction, SSRIs (standard antidepressants like fluoxetine, sertraline, and paroxetine) have anti-inflammatory effects beyond their effects on serotonin. They reduce circulating cytokines including IL-1beta and CRP in people with major depression (Osimo et al., 2019). Paroxetine, specifically, has been shown in preclinical IBD models to reduce gut inflammation, restore butyrate-producing bacteria, and rebuild gut lining markers like Mucin 2 (Ning et al., 2023; Tang et al., 2015). The drug was designed for the brain and it is acting on the gut.
Probiotics have also shown effects, though with more caveats. A specific combination of Lactobacillus helveticus R0052 and Bifidobacterium longum R0175 reduced psychological stress and depression scores in clinical trials (Messaoudi et al., 2011), and was associated with increased BDNF levels in people with major depressive disorder when used as adjunctive therapy (Kazemi et al., 2019). The key word is specific. Generic probiotic supplements are not equivalent to evidence-backed strains, and strain-level precision matters.
The Bigger Picture of the Gut-Brain Axis
What the IBD research makes clear is something that probably applies to a lot more than IBD. When chronic inflammation, chronic pain, mood symptoms, and gut dysfunction show up together (and they often do), treating each one in isolation will only ever capture part of the problem.
This is the reason integrative frameworks are so important when treating a person; a person is a whole system (of systems!). A system that runs in loops needs to be addressed at multiple points in the loop, because addressing one point alone gets undone by the others.
It is also why the relationship between a patient and their care matters so much. Chronic, self-reinforcing conditions do not respond well to a single intervention delivered once. They respond to consistent attention, adjustment, and support across time.
The research on IBD and depression is not a niche finding. It is a window into how a lot of chronic illness actually works.
Source
Simoes, J. L. B., Braga, G. C., Assmann, C. E., and Bagatini, M. D. (2026). Targeting the gut-immune-brain axis: pharmacological insights from depression in inflammatory bowel disease. Frontiers in Pharmacology, 17. https://doi.org/10.3389/fphar.2026.1793292